While there are many great tools and technologies available to support drug development, the number of platforms used for CDx development remains very limited. Looking at the FDA CDx approvals website as of April 2020, here is a summary of the technologies and platforms with approved diagnostics:
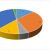
PCR and IHC remain the most popular approaches, followed by NGS and FISH or ISH. This is even further skewed in that many of these tests are for the same target (eg, multiple tests for HER2 and EGFR mutations) with multiple manufacturers developing tests for the same target. Overall, there are few platforms represented in the sum of all approved CDx. These numbers are reflected in the CDx that NeoGenomics has in early phases of development, with a strong emphasis on IHC. These are all fine tools and approaches, but for the most part not cutting edge.
Generally, there is a preference for well-characterized approaches and technologies for regulated tests. There is an infrastructure of well-established IVD partners to support GMP kit development, regulatory approval, marketing and commercialization. Requirements for regulatory approval are generally well understood. Finally, IVD tests need to be compatible with the existing ecosystem of platforms used as hospital and commercial laboratories. These factors make technologies like PCR and IHC the path of least resistance to get a CDx to market.
This does not mean that CDx development is written in stone, forever and inextricably linked to these few technologies. NeoGenomics has a few CDx tests outside of this spectrum under development or consideration, in very early phases of development. New approaches, including flow cytometry, multiplex-immunofluorescence, and RNA counting are up-and-coming. The first of these to get to market represents a steeper climb for the developer, relative to IHC or PCR, but not an impossible climb. We’re excited to be working with these technologies at earlier phases of development, and look forward to potentially bringing them to market as approved devices.

