Where trust meets results - 300+ biopharma partners
At NeoGenomics, we understand the needs of pharmaceutical and biotechnology companies, and we align with our partners to support development of therapies that will save lives. With over a decade of experience, we are a trusted partner in oncology research and development, delivering solutions to advance your drug development and clinical trial programs for targeted therapies and emerging treatment modalities.
No matter the scale or business need, we deliver:
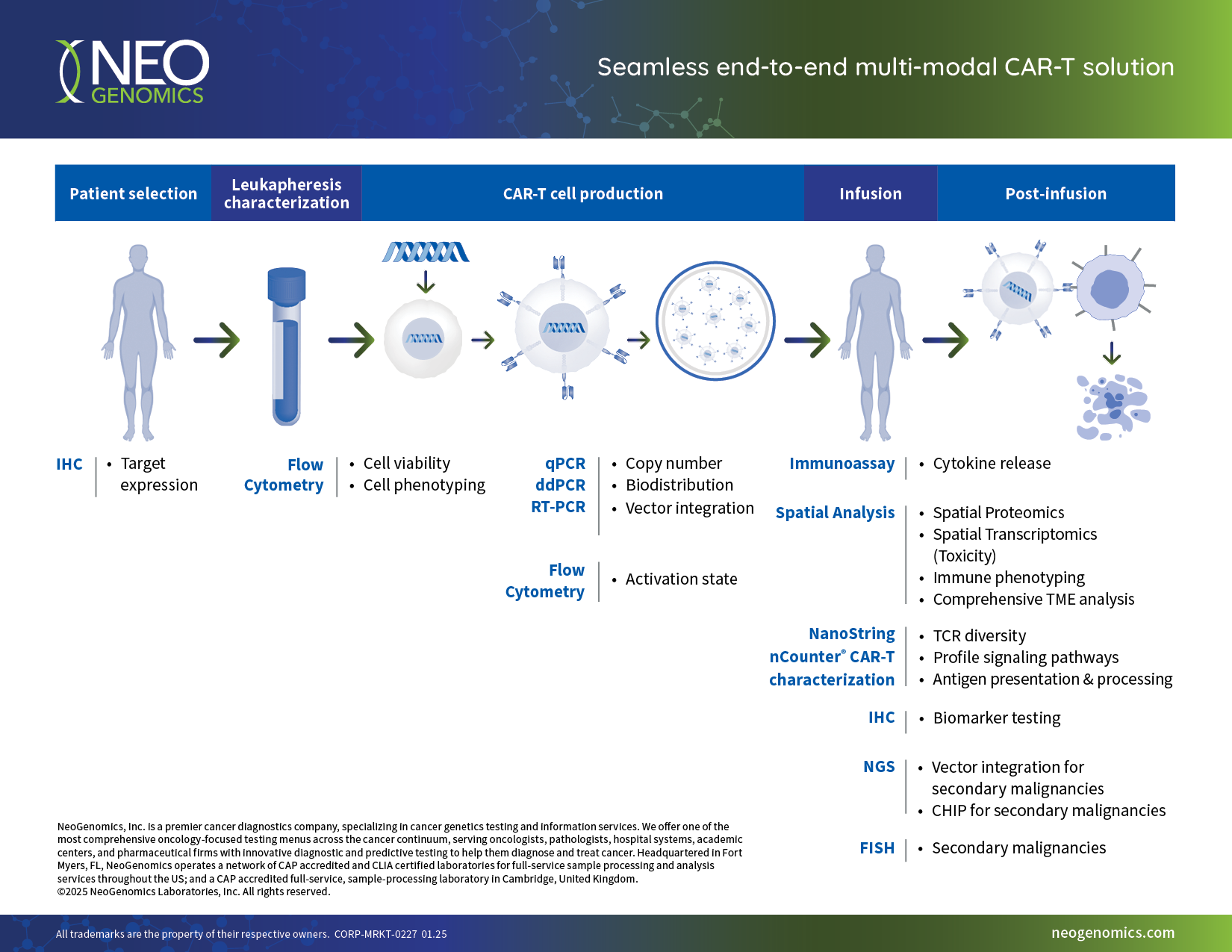
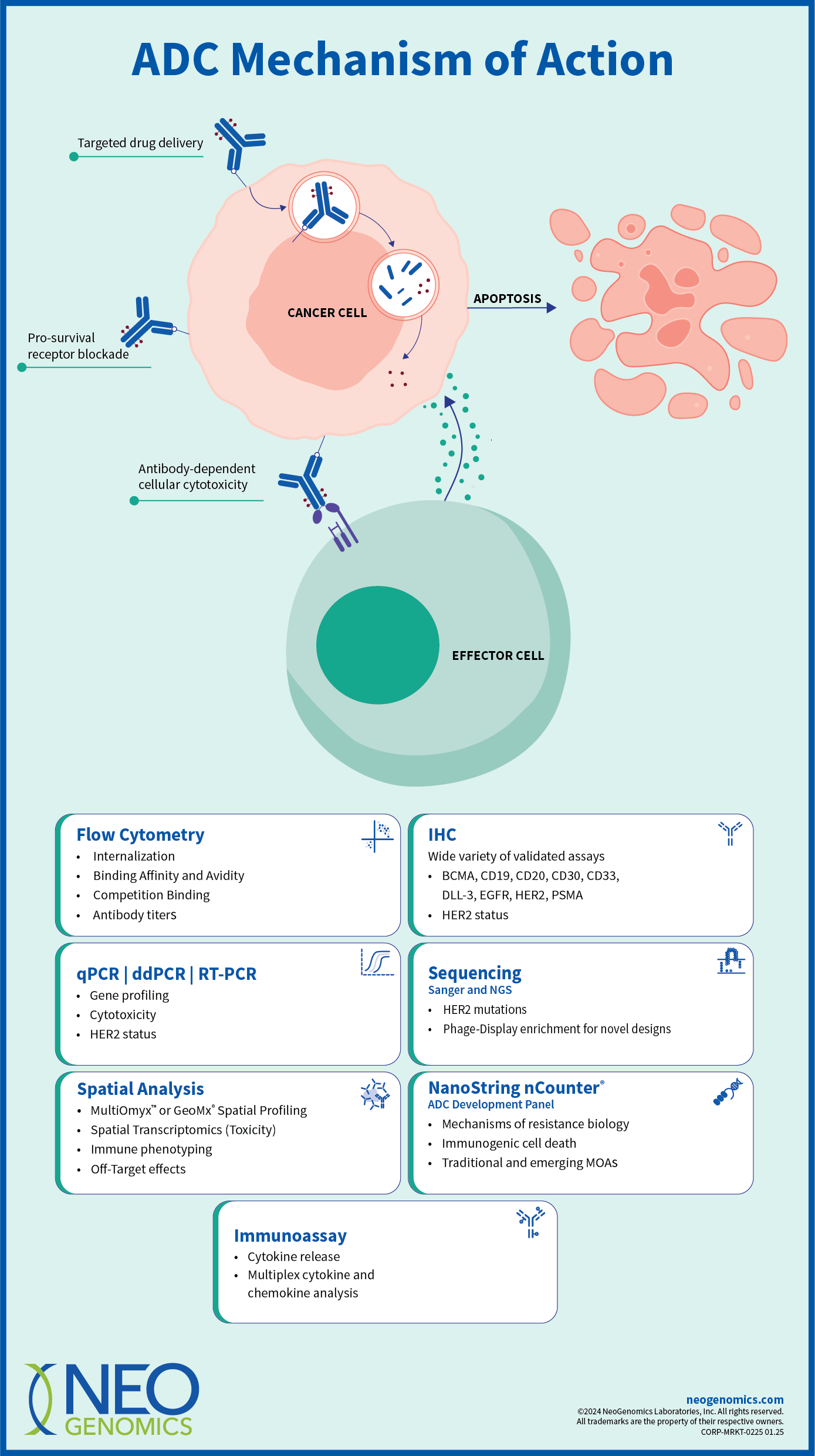
- Seamless end-to-end multimodal testing services, eliminating the need for multiple vendors
- Broad biopharmaceutical test menu and integrated data analysis
- Advanced customized solutions to accelerate your development program
- Deep scientific and technical expertise with MDs, PhDs, and dedicated project managers ensuring project integrity
- In-house regulatory and compliance team to effectively navigate complex regulatory processes and support submissions
- Certified pathology staff capable of performing US FDA-approved companion diagnostic confirmations
- Large-scale, real-world oncology data to better inform clinical trials
Move your drug development and clinical trials forward, faster